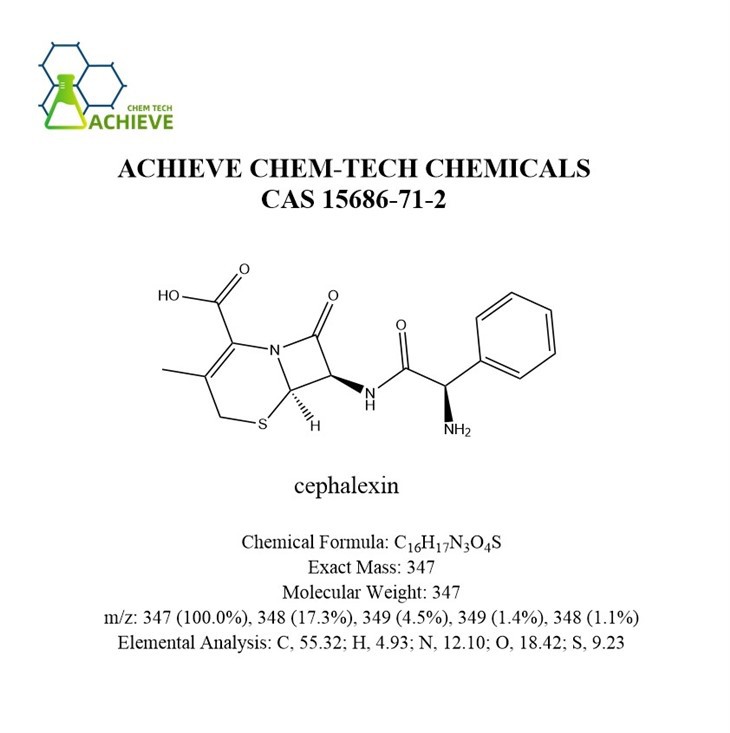
Shaanxi BLOOM Tech Co., Ltd. is one of the most experienced manufacturers and suppliers of cephalexin powder cas 15686-71-2 in China. Welcome to wholesale bulk high quality cephalexin powder cas 15686-71-2 for sale here from our factory. Good service and reasonable price are available.
Cephalexin powder, usually appears as a white to slightly yellow crystalline powder, and its slightly foul odor provides a simple way for users to identify it. CAS 15686-71-2, The molecular formula C16H17N3O4S shows significant differences in solubility in different solvents. In water, it can only dissolve slightly, which means that it takes some time and conditions to fully dissolve in water. Cefalexin is insoluble in ethanol, chloroform, or ether. It is a semi synthetic first generation oral cephalosporin antibiotic that can inhibit the synthesis of cell walls, causing cell contents to expand and dissolve, killing bacteria. It has a certain antibacterial effect on bacteria such as Escherichia coli, Proteus mirabilis, and Salmonella, and can be used to treat urinary tract infections, urethritis, chronic cystitis, and other urinary system infections. It has anti-inflammatory effects and can be used to alleviate symptoms such as skin itching, pain, and burning caused by bacterial infections.

|
Chemical Formula |
C16H17N3O4S |
|
Exact Mass |
347 |
|
Molecular Weight |
347 |
|
m/z |
347 (100.0%), 348 (17.3%), 349 (4.5%), 349 (1.4%), 348 (1.1%) |
|
Elemental Analysis |
C, 55.32; H, 4.93; N, 12.10; O, 18.42; S, 9.23 |
|
|
|

Molecular Structure and Functional Groups
The chemical structure of cefaclor contains a β - lactam ring, which is a key component for its antibacterial activity. The β - lactam ring has unique chemical properties and is a quaternary ring structure with high reactivity between the nitrogen atom and carbonyl carbon atom on the ring. This structure enables cefaclor to bind to the key enzyme in bacterial cell wall synthesis, penicillin binding proteins (PBPs), thereby inhibiting the synthesis of bacterial cell walls, causing bacterial cell contents to expand and dissolve, ultimately killing bacteria.
In addition to the β - lactam ring, cephalosporin molecules also contain functional groups such as amino, carboxyl, and sulfur atoms. Amino groups have alkalinity and can react with acids to form salts; Carboxyl groups have acidity and can react with bases to form salts; Sulfur atoms participate in some important chemical reactions, such as redox reactions. The presence of these functional groups endows cephalosporin with certain chemical activity and enables it to participate in various chemical reactions.

Stability

Stability to heat
Cephalexin powder is relatively stable in a dry state, but undergoes decomposition reactions at higher temperatures. Studies have shown that when the temperature rises to a certain level, the β - lactam ring of cefaclor undergoes a ring opening reaction, leading to the loss of its antibacterial activity. For example, under heating conditions, cephalosporin may undergo molecular rearrangement, hydrolysis, and other reactions, generating some inactive products. Therefore, in the preparation, storage, and use of cefaclor, it is necessary to strictly control the temperature conditions to avoid the impact of high temperatures on it.
Stability of strong acids and bases
Cefalexin is unstable in strong acid and strong alkali environments. In a strong acidic environment, cephalosporin undergoes hydrolysis reaction, opening the β - lactam ring to generate corresponding carboxylic acids and amine compounds. In a strong alkaline environment, the lactam ring may decompose, and other functional groups in the molecule may also react, leading to changes in the chemical structure of the drug and a decrease in its efficacy. Therefore, when preparing ceftriaxone solution, it is necessary to choose an appropriate pH range to ensure its stability.


Stability to ultraviolet radiation
Due to the high energy of ultraviolet radiation, it may break the chemical bonds of cefaclor molecules, leading to their decomposition. Ultraviolet irradiation can break certain chemical bonds in the molecule of cefaclor, producing active intermediates such as free radicals. These intermediates further react, leading to the degradation of the drug. Therefore, when storing and using ceftriaxone, it is necessary to avoid direct sunlight and ultraviolet radiation, and dark packaging and storage methods can be used.
Chemical reactivity
Cefalexin undergoes hydrolysis reaction in solution, especially under acidic or alkaline conditions. The hydrolysis reaction can cause the opening of the β - lactam ring, resulting in the formation of carboxylic acids and amine compounds. The rate of hydrolysis reaction is influenced by factors such as temperature, pH value, and solution concentration. Generally speaking, the higher the temperature, the more neutral the pH value, and the higher the solution concentration, the faster the hydrolysis reaction rate. For example, under acidic conditions, the hydrolysis reaction of cefoperazone is accelerated, producing corresponding carboxylic acids and products such as phenylglycine methyl ester hydrochloride.
The sulfur atoms and other functional groups in cephalosporin molecules have a certain degree of reducibility and are prone to oxidation reactions. Under the action of oxidants, sulfur atoms may be oxidized, leading to changes in the chemical structure of the drug. Oxidation reaction can darken the color of cefaclor and also affect its antibacterial activity. For example, under certain oxidative conditions, ceftriaxone may generate sulfoxides or sulfone compounds, which typically have lower antibacterial activity than ceftriaxone itself.
Cefalexin can undergo a chemical reaction with ninhydrin solution, resulting in a color change. This color reaction is helpful for the qualitative or quantitative detection of cefaclor. Indene ketone can react with functional groups such as amino groups in cephalosporin molecules to produce purple or blue compounds. The content of cefaclor can be determined by measuring the absorbance of the reaction product. This reaction has important application value in drug analysis and can be used for drug quality control and clinical testing.
Chemical properties related to solubility
Solubility in different solvents
Cefalexin is slightly soluble in water and insoluble in ethanol, chloroform, or ether. This solubility characteristic is related to its molecular structure and functional groups. Water is a polar solvent, and the polar functional groups (such as carboxyl and amino groups) in cephalosporin molecules can form hydrogen bonds with water molecules, thereby giving it a certain solubility in water. However, organic solvents such as ethanol, chloroform, and ether have lower polarity and weaker interactions with cephalosporin molecules, resulting in lower solubility of cephalosporin in these solvents.
The influence of solubility on formulation preparation
Due to the solubility characteristics of cefaclor, special methods are required when preparing its formulations. For example, when preparing oral liquid formulations, some cosolvents or solubilizers such as propylene glycol, polyethylene glycol, etc. can be added to increase the solubility of cefaclor in water. When preparing solid formulations, it is necessary to control the size and shape of particles to improve their dissolution rate and bioavailability.
High Strength
We offer a variety of transmission components, including sprockets, roller chains, gears, couplings, racks, hubs, pulleys, taper sleeves, bearing seats, and more.
Professional
We offer a variety of transmission components, including sprockets, roller chains, gears, couplings, racks, hubs, pulleys, taper sleeves, bearing seats, and more.
Interaction with drugs

Interaction with Probenecid
The simultaneous use of cefaclor and propiconazole can delay the renal excretion of cefaclor and increase the blood concentration of cefaclor. Probenecid can competitively inhibit the secretion of cefoxil by renal tubules, thereby reducing the excretion of cefoxil. But there are also reports suggesting that probenecid can increase the excretion of cefaclor in bile. This interaction may affect the efficacy and safety of cefaclor, so caution should be exercised when using combination therapy.
Interaction with cholesteryl amine (cholesteryl amine)
The co administration of cefaclor and cephalexin powder can reduce the average peak blood concentration of cefaclor. Kolacetamide is an anion exchange resin that can bind with cefoxil to form insoluble complexes, thereby reducing the absorption of cefoxil in the intestine. Therefore, when using both drugs simultaneously, it is necessary to adjust the dosage or administration time of cefaclor.


Interaction with aminoglycoside drugs
The combination of cefaclor and aminoglycoside drugs can increase nephrotoxicity. Aminoglycoside drugs themselves have a certain degree of nephrotoxicity, and ceftriaxone may also cause a certain burden on the kidneys after metabolism in the body. When two drugs are used in combination, the risk of nephrotoxicity increases. Therefore, close monitoring of the patient's renal function is necessary when using combination therapy.
Interaction with live typhoid vaccine
The co administration of cefaclor and typhoid live vaccine may reduce the immune response of typhoid live vaccine. The possible mechanism is that cefaclor has antibacterial activity against Salmonella typhi, which can inhibit the growth and reproduction of Salmonella typhi in vaccines, thereby affecting the immune efficacy of vaccines. Therefore, cephalosporin should be avoided for a period of time before and after the use of live typhoid vaccine.

Metabolic related chemical properties

Metabolic pathways
Cefalexin is mainly metabolized in the liver and excreted through the kidneys. In the liver, ceftriaxone undergoes a series of enzymatic reactions, such as oxidation, reduction, hydrolysis, etc., to generate some metabolites. These metabolites may have different pharmacological activities and toxicity. For example, metabolites of cephalosporin may include deacetylated cephalosporin, 7-aminocephalosporanic acid, etc.
The impact of metabolism on drug efficacy and safety
The metabolic process of cefaclor affects its efficacy and safety. Different individuals have varying metabolic abilities towards cefaclor, which may result in different concentrations and duration of action of the drug in the body. For example, individuals with strong metabolic abilities may metabolize cefuroxime into inactive products faster, thereby reducing the efficacy of the drug; Individuals with weaker metabolic abilities may accumulate higher drug concentrations in their bodies, increasing the risk of adverse drug reactions.

Chemical properties related to antibacterial spectrum
Antibacterial spectrum range
The antibacterial spectrum of Cefotaxime is similar to that of Cefotaxime, but its antibacterial activity is inferior to the latter. Except for Enterococcus and methicillin-resistant Staphylococcus, Gram positive cocci are sensitive to cefoxil. Cefalexin has a good antibacterial effect on Neisseria, but is less sensitive to influenza bacteria. It has a certain antibacterial effect on some Escherichia coli, Proteus mirabilis, Klebsiella pneumoniae, and Salmonella. Other Escherichia coli, Acinetobacter baumannii, Pseudomonas aeruginosa, and Bacteroidetes fragilis are resistant to Cefalexin.
The relationship between antibacterial mechanism and chemical structure
The antibacterial effect of cefaclor is achieved by binding to one or more penicillin binding proteins (PBPs) in bacteria, inhibiting cell wall synthesis during bacterial cell division. The β - lactam ring in its molecular structure is a key site for binding with PBPs. Different bacteria have different types of PBPs, and the affinity of cefaclor to different PBPs varies, which determines the differences in its antibacterial spectrum. For example, ceftriaxone has a strong affinity for PBPs of Gram positive bacteria, thus exhibiting good antibacterial activity against Gram positive bacteria; However, some PBPs have weak affinity for Gram negative bacteria, so their antibacterial activity against some Gram negative bacteria is relatively poor.
Toxicity related chemical properties
Acute toxicity
Cephalexin powder has low acute toxicity, but it may still cause damage to the body at high doses. Animal experiments have shown that the median lethal dose (LD50) of orally administered ceftriaxone in rats is relatively high, indicating that its acute toxicity to animals is relatively low. However, in clinical applications, if cephalosporin is ingested or overused, it may cause a series of adverse reactions such as nausea, vomiting, diarrhea, rash, etc.
Chronic toxicity
Long term use of cefaclor may cause some chronic toxic reactions. For example, long-term medication can lead to dysbiosis of the microbiota and the occurrence of secondary infections. This is because while cephalosporin inhibits pathogenic bacteria, it also affects the growth and balance of normal gut microbiota, leading to the proliferation of some drug-resistant or opportunistic pathogens and causing new infections. In addition, long-term use of cefaclor may also lead to deficiencies in vitamin K and vitamin B.
Mutability, teratogenicity, and carcinogenicity
At present, there is relatively little research on the mutagenicity, teratogenicity, and carcinogenicity of cefepime. Some in vitro and animal experiments have shown that ceftriaxone may have certain mutagenicity under certain conditions, but its significant teratogenic and carcinogenic effects have not been found in clinical applications. However, in order to ensure medication safety, special populations such as pregnant women, breastfeeding women, and children need to carefully weigh the pros and cons when using cefaclor.
Hot Tags: cephalexin powder cas 15686-71-2, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 4 bromo 2 chlorospiro fluorene 9 9 xanthene , pure aspirin powder, CAS 1115639 92 3, Solvent, propylene oxide market, CAS 1936437 58 9