4-Nitroquinoline N-oxide(4-NQO) is an organic compound with the chemical formula C9H6N2O4, CAS 56-57-5, and appears as a light yellow to yellow solid. It can dissolve in organic solvents such as acetic acid, acetone, chloroform, and dichloromethane, but is insoluble in water. This compound has a complex molecular structure, including a nitrogen oxide group and a quinoline group. There are two nitro substituents in the molecule, located at the 4 and N positions of the quinoline ring. Synthesized through various methods, such as diazotization reaction, oxidation reaction, etc. One common synthesis route is to use 4-hydroxyquinoline as raw material, introduce nitro groups through diazotization reaction, and then oxidize with oxidant to obtain the target compound. In addition, the compound can also be prepared by direct nitration of quinoline. It has a wide range of applications and applications, including research on anti-cancer drugs, fluorescent dyes, photosensitizers, synthetic intermediates, analytical reagents, and research tools for bioactive molecules.

|
|
|
|
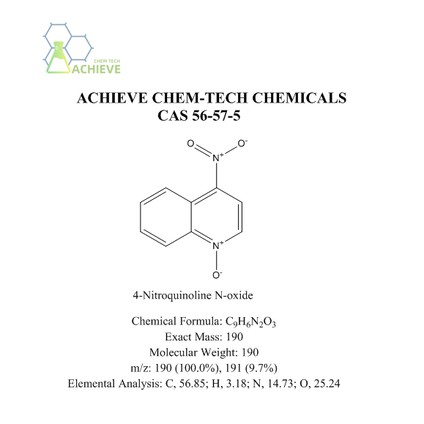
Chemical Formula |
C9H6N2O3 |
|
Exact Mass |
190 |
|
Molecular Weight |
190 |
|
m/z |
190 (100.0%), 191 (9.7%) |
|
Elemental Analysis |
C, 56.85; H, 3.18; N, 14.73; O, 25.24 |

4-Nitroquinoline N-oxide is a commonly used chemical substance in genetic toxicity research, with multiple mechanisms of action and applications.

1. Carcinogenic studies:
4-NQO is widely used in the study of carcinogenic effects. It is a strong carcinogen that can induce tumor formation in various animal models and human cells. In the process of studying carcinogenic effects, 4-NQO is often used as a model compound to investigate its mechanism of inducing tumors and its synergistic effects with other compounds. In addition, 4-NQO can also be used to explore the molecular mechanisms of tumor occurrence and search for effective treatment strategies.
2. Research on gene expression regulation:
4-NQO can affect the regulation process of gene expression, mainly through binding to DNA and affecting transcription factor activity. In gene expression regulation research, 4-NQO is used as a model compound to investigate the regulatory mechanisms and influencing factors of gene expression. By using 4-NQO to treat cells or tissues, researchers can study changes in gene expression and its relationship with tumor development. In addition, 4-NQO can also be used to screen and evaluate the potential effects of drugs or other compounds on gene expression.


3. Signal transduction research:
4-NQO can affect various signal transduction pathways, including MAPK, PI3K/Akt, NF- κ Class B. In signal transduction research, 4-NQO is used as a tool compound to investigate the mechanisms and regulation of signal transduction. By using 4-NQO to treat cells or tissues, researchers can investigate the activation of signal transduction pathways and their relationship with tumor development. In addition, 4-NQO can also be used to screen and evaluate the potential effects of drugs or other compounds on signal transduction.
4. Immunotoxicity research:
4-NQO has certain immunotoxicity and can affect the activity and function of immune cells. In immunotoxicity studies, 4-NQO is used as a model compound to investigate changes and regulation of the immune system. By using 4-NQO to treat immune cells or whole animals, researchers can study the apoptosis, activation of immune cells and their relationship with tumor development. In addition, 4-NQO can also be used to screen and evaluate potential immunomodulatory drugs or other compounds for their impact on the immune system.

4-Nitroquinoline N-oxide (4-NQO) is an important chemical substance that plays a crucial role in genetic toxicity research. It not only has multiple mechanisms of action, but is also widely used in fields such as cancer research, organic synthesis, and drug development.
Chemical properties
It is an organic compound with the molecular formula C9H6N2O3 and a molecular weight of approximately 190.16 (slightly different literature, such as 192.1636). It usually exists in the form of yellow to brown crystals or powders, with a high melting point (154-156 ° C) and boiling point (387.64 ° C). In addition, the density of 4-NQO is about 1.42 g/cm ³, and the refractive index is about 1.5570. 4-NQO is soluble in organic solvents such as acetone and is sensitive to light. It should be stored at -20 ° C to avoid decomposition.
The chemical structure of 4-NQO contains nitro (NO2) and N-oxide groups, which play critical roles in chemical reactions. The nitro group makes 4-NQO a strong oxidant that can participate in various redox reactions. The N-oxide group gives it unique biological activity, especially in terms of genetic toxicity.
Mechanism of action
4-NQO exerts its genotoxic effects in organisms through various mechanisms. These mechanisms mainly include DNA damage, oxidative stress, and inhibition of DNA synthesis.
Oxidative stress refers to a pathological state that occurs when there is excessive production of ROS in an organism and the antioxidant defense system is unable to effectively eliminate it. 4-NQO further exacerbates DNA damage by inducing oxidative stress. ROS can not only directly attack DNA, but also cause membrane lipid peroxidation, protein oxidation, and mitochondrial dysfunction. These effects collectively exacerbate the oxidative stress state of cells, making them more susceptible to genetic toxicity.
In addition to directly damaging DNA, 4-NQO can also affect cell proliferation and differentiation by inhibiting DNA synthesis. Research has shown that 4-NQO can inhibit the activity of DNA polymerase, thereby slowing down the elongation rate of DNA strands. This inhibitory effect not only hinders DNA replication, but may also affect the DNA repair process, making cells more susceptible to genetic damage.
Application
4-NQO has broad application value in multiple fields, especially in cancer research, organic synthesis, and drug development.
Cancer Research
4-NQO is an effective carcinogen commonly used in cancer modeling and mechanism research. It can induce cellular carcinogenesis through mechanisms such as DNA damage and oxidative stress. In animal experiments, 4-NQO has been widely used to induce the occurrence of cancers such as oral cancer and tongue mucosal squamous cell carcinoma. These models not only help to reveal the pathogenesis and molecular basis of cancer, but also provide important experimental evidence for cancer prevention and treatment.

Related research
4-NQO, as an important chemical substance, has received widespread attention and research in both academia and industry. Here are some research progress and achievements related to 4-NQO.

(1) 4-NQO induced DNA damage and repair
Research has shown that 4-NQO can induce various types of DNA damage, including DNA strand breaks, base damage, and DNA protein cross-linking. These damages can lead to replication errors and abnormal gene expression during DNA replication and transcription processes. However, cells have a complex DNA repair mechanism that can recognize and repair these damages. Research has shown that DNA damage induced by 4-NQO is mainly repaired through the nucleotide excision repair (NER) pathway. NER is a highly conserved DNA repair mechanism that can recognize and remove damaged DNA fragments, and perform precise repairs.
(2) The carcinogenic mechanism of 4-NQO
The carcinogenic mechanism of 4-NQO has always been a hot research topic. Research has shown that 4-NQO can induce mechanisms such as DNA damage and oxidative stress, leading to cellular gene mutations and chromosomal abnormalities, thereby increasing the risk of cellular carcinogenesis. In addition, 4-NQO can also affect the proliferation and differentiation processes of cells, making them more susceptible to genetic damage. These mechanisms collectively explain why 4-NQO is an effective carcinogen in animal experiments.


(3) Application of 4-NQO in drug development
4-NQO has broad application prospects in drug development. By studying the effects of 4-NQO on cell proliferation, differentiation, and apoptosis, compounds with anti-tumor activity can be screened. These compounds have significant value in the development of anticancer drugs. In addition, 4-NQO can also serve as a model compound for drug metabolism and toxicity studies, used to evaluate the metabolic pathways and toxicity mechanisms of drugs. These studies not only help to reveal the efficacy and safety of drugs, but also provide important experimental evidence for the clinical application of drugs.
(4) Environmental and Health Effects of 4-NQO
With the accelerated development of industrialization and urbanization, the pollution problem of chemical substances such as 4-NQO in the environment is becoming increasingly prominent. Research has shown that 4-NQO can enter the environment and organisms through various pathways, posing potential threats to the environment and human health. Therefore, studying the migration, transformation, and bioaccumulation processes of 4-NQO in the environment, as well as its toxic effects and ecological risks on organisms, is of great significance. These studies not only contribute to the development of scientifically sound environmental protection policies and measures, but also provide important scientific basis for safeguarding human health and environmental safety.


There are many synthesis methods for 4-Nitroquinoline N-oxide, and the two most common ones will be listed in the following text.
Method 1:
Synthesis of 4-Methoxyquinoline-N-Oxide
React 4-hydroxyquinoline with sodium methoxide to obtain 4-methoxyquinoline-N-oxide.
C9H7NO + CH3OH + NaOH → C9H7NO-N oxide + H2O + NaCl
This reaction is a typical alcoholization reaction, in which sodium methoxide is used as an alcoholizing agent to methylate the hydroxyl group of 4-hydroxyquinoline-N-oxide.
Synthesis of 4-chloroquinoline-N-oxide
4-Methoxyquinoline-N-oxide was treated with acetyl chloride to obtain 4-chloroquinoline-N-oxide.
C9H7NO-N-Oxide + CH3COCl → 4-Chloroquinoline-N-Oxide + HCl
This reaction is an acyl chlorination reaction, in which acetyl chloride acts as an acyl chlorinating agent to acetylate the methoxy group of 4-methoxyquinoline-N-oxide to obtain 4-chloroquinoline-N-oxide.
Synthesis of 4-aminoquinoline-N-oxide
Palladium catalyzed hydrogenation of 4-chloroquinoline-N-oxide in liquid ammonia yields 4-aminoquinoline-N-oxide.
4-Chloroquinoline-N-Oxide + H2 + Pd/C → 4-aminoquinoline-N-Oxide
This reaction is a typical catalytic hydrogenation reaction, using palladium as a catalyst to replace the chlorine atom of 4-chloroquinoline-N-oxide with a hydrogen atom to obtain 4-aminoquinoline-N-oxide.
Synthesize the target product
Under acidic conditions, the target product 4-NQO was synthesized by nitrating quinoline-N-oxide with sulfuric acid and nitric acid.
4-Aminoquinoline-N-Oxide + H2SO4 + HNO3 → C9H6N2O3 + H2O + SO2 + NO2
This reaction is a typical nitration reaction, in which sulfuric acid and nitric acid are used as nitrating agents to nitrate the amino group of 4-aminoquinoline-N-oxide, resulting in the target product. Simultaneously producing by-products such as sulfuric acid, nitrogen dioxide, and water.
The above are the detailed steps of Method One and its corresponding chemical reaction formula. It should be noted that these reaction conditions and product purity may vary depending on the experimental conditions and purity requirements of the target product. In practical operation, it is recommended to adjust and optimize according to specific experimental conditions and purity requirements of the target product.

Method 2:
Dissolve the raw materials
Dissolve 5-hydroxyindole in ethanol to obtain an ethanol solution of 5-hydroxyindole. This step is preparatory work aimed at dissolving 5-hydroxyindole in ethanol for use in subsequent reactions.
Synthesize 4-bromoquinoline
In the presence of sodium ethanol, benzyl bromide was added to an ethanol solution of 5-hydroxyindole, heated to a certain temperature, kept stirring, and reacted for a period of time to obtain 4-bromoquinoline. The reaction equation for this step can be expressed as:
C8H7NO + CH3CHBrNa → C9H6BrN + NaOH + H2O
This reaction is a substitution reaction, where sodium ethanol acts as a base to help 5-hydroxyindole remove hydroxyl groups and react with benzyl bromide to produce 4-bromoquinoline.
Synthesize 4-Morpholinoanine
React 4-bromoquinoline with morpholine at a certain temperature and pressure, and separate the generated 4-Morpholinoaniline through column chromatography or other separation methods. The reaction equation for this step can be expressed as:
C9H6BrN + C4H9NO → C10H14N2O + NaOH + H2O
This reaction is a substitution reaction, in which morpholine acts as a substituent and reacts with 4-bromoquinoline to form 4-Nitroquinoline N-oxide.
4-Nitroquinoline N-oxide, once a obscure chemical compound, has become indispensable in biomedical research and toxicology. Its dual role as a mutagen and oxidative stress inducer provides a unique lens through which to study cancer biology, DNA repair, and drug development. While its carcinogenic potential necessitates rigorous safety protocols, ongoing innovations in detection and therapy continue to expand its applications. As science advances, 4-NQO will undoubtedly remain a vital tool in unraveling the complexities of disease and discovering novel interventions.
Frequently Asked Questions
What is n-methylmorpholine n-oxide?
+
-
N-methylmorpholine N-oxide (N MMO) is defined as a cyclic N-oxide that is particularly effective for dissolving cellulose and is a key component in the lyocell industry for producing regenerated cellulose fibers.
What is 4-methylmorpholine used for?
+
-
4-methylmorpholine, also known as N-Methylmorpholine, is a cyclic tertiary amine and is commonly used as a highly valuable solvent and a fine chemical for cellulose.
Is 4 methyl morpholine toxic?
+
-
May be moderately toxic by ingestion, inhalation and skin absorption. Very irritating to skin, eyes, and mucous membranes. Used as a solvent and to make pharmaceuticals.
Hot Tags: 4-nitroquinoline n-oxide cas 56-57-5, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, 2 5 Dihydroxybenzaldehyde, BENZENE D6, 3 Nitrobenzaldehyde 99 , gs 441524 remdesivir, N N N Trimethylethylenediamine, Methylamine hydrochloride powder