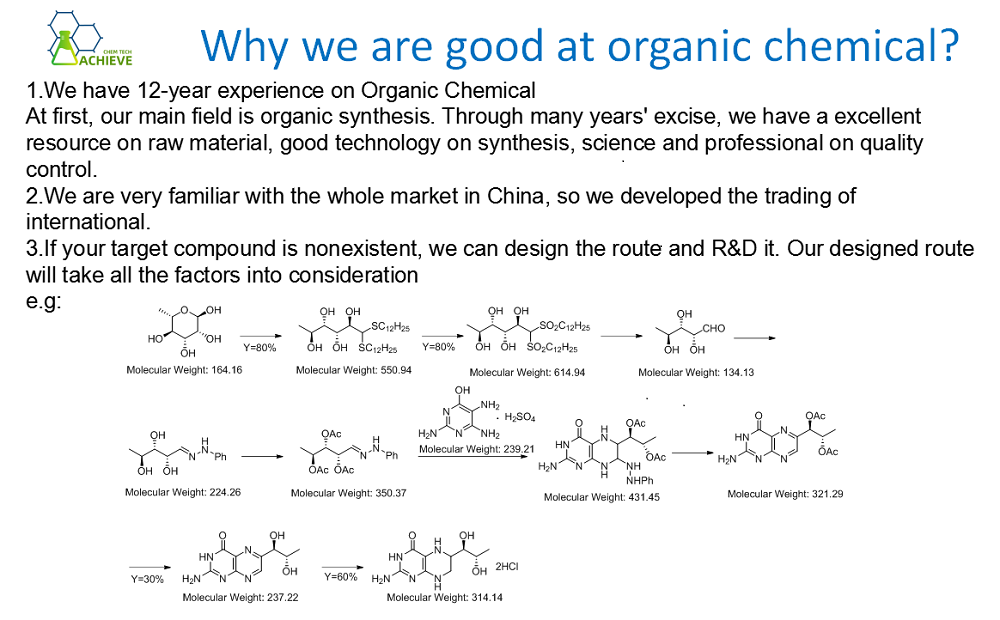
Tetrabutylammonium bromide powder, also known as tetrabutylammonium bromide, is an organic salt, CAS 1643-19-2, The molecular formula is C16H36BrN. Pure products are white crystals or powders, with deliquescence, a special odor, and good stability at room temperature and pressure. Easy to dissolve in water, ethanol, ether, and acetone, slightly soluble in benzene. Toxic, commonly used as an intermediate in organic synthesis, phase transfer catalyst, and ion pair reagent. It is used for the synthesis of pharmaceutical intermediates such as benzyltriethylammonium chloride, ethyl cinnamate, pseudoionone, etc., as well as for the synthesis of organic drugs such as anti infective drugs such as bamacillin and sultamicillin. It is also used as a catalyst in the synthesis of listed drugs such as anti diabetes drug dagelin with a new mechanism of action. In addition, it can not only be used as a phase transfer agent to synthesize tiopronin, a liver function improvement and metabolic antidote, but also can be used as a bromine agent to participate in the synthesis of muscle relaxant reversal agent, Shugenglucuronium.

|
Chemical Formula |
C16H36BrN |
|
Exact Mass |
321 |
|
Molecular Weight |
322 |
|
m/z |
321 (100.0%), 323 (97.3%), 322 (17.3%), 324 (16.8%), 325 (1.2%), 323 (1.1%) |
|
Elemental Analysis |
C, 59.61; H, 11.26; Br, 24.79; N, 4.34 |
|
|
|

Tetrabutylammonium bromide (TBAB), as an important quaternary ammonium salt compound, has shown wide application value in various fields such as chemical synthesis, pharmaceutical manufacturing, environmental protection, electrochemistry, and materials science due to its unique chemical structure (molecular formula C ₁₆ H ∝₆ BrN) and physical properties (white crystals, easy solubility in water and organic solvents, deliquescence).
1. Phase transfer catalyst (PTC)
The most classic use is as a phase transfer catalyst, used to promote the transfer and reaction of substances in two-phase reaction systems (such as water organic phases). The mechanism of action is that the quaternary ammonium cation (C ₄ H ₉) ₄ N ⁺) can combine with anions in the aqueous phase (such as OH ⁻, Br ⁻) to form ion pairs and transfer to the organic phase, thereby breaking the phase separation state of reactants in different solvents and significantly improving the reaction rate and yield.
Typical application cases:
Pseudo violet ketone synthesis: Tetrabutylammonium bromide powder as a catalyst can promote the condensation reaction of citral and acetone in the synthesis of pseudo violet ketone, with a yield increase of over 85%, far higher than the traditional process's 60%.


Cinnamic acid decarboxylation and bromination: TBAB is combined with 2-iodoylbenzoic acid (IBX) to achieve efficient decarboxylation and bromination of cinnamic acid in a mixed solvent of acetonitrile and water, with a yield of 92% and mild reaction conditions (60 ℃, atmospheric pressure).
Synthesis of anti infective drugs: TBAB is used to catalyze the synthesis of β - lactam antibiotics such as ampicillin and sultamicillin, by promoting the formation of carbon nitrogen bonds, shortening reaction time, and improving product purity.
2. Alkylation and Acylation Reactions
The quaternary ammonium salt structure of TBAB makes it an ideal catalyst for alkylation and acylation reactions. For example:
N-alkylation reaction: Tetrabutylammonium bromide can catalyze the N-alkylation reaction of 3-indole with bromoalkane to generate N-alkylindole derivatives, which have important value in drug synthesis.
Alkylation of enolates: TBAB reacts with enolates to form stable alkylated products, which are used for the synthesis of steroid compounds and natural products.
3. Green chemistry process
With the increasingly strict environmental regulations, TBAB is highly favored in green chemistry due to its low toxicity, high purity, and recyclability. For example:
Unsolvented bromination reaction: By introducing bromine vapor into TBAB solid, tributylammonium bromide (TBABr ∝) can be directly synthesized with a yield of 84%, avoiding the volatilization pollution of bromine in traditional bromination processes.
Synthesis by ion exchange method: use gel type strong basic anion exchange resin to replace Br ⁻ in TBAB solution with OH ⁻ to generate tetrabutylammonium hydroxide (TBAOH). The resin is renewable and recycled, significantly reducing production costs.

The dual contribution of antibacterial and drug synthesis in the field of pharmaceutical manufacturing

1. Antibacterial drug raw materials
TBAB itself has strong bactericidal ability and can be directly used to prepare antibacterial disinfectants. Its mechanism of action is to destroy the permeability of bacterial cell membranes, leading to leakage of cellular contents, thereby killing bacteria, fungi, and viruses.
Application scenarios:
Medical facility disinfection: TBAB solution is used for disinfection of surgical instruments and ward environment, effectively killing drug-resistant bacteria such as methicillin-resistant Staphylococcus aureus (MRSA).
Food processing plant sterilization: TBAB, as a residue free fungicide, is used for cleaning meat and dairy processing equipment to ensure food safety.
2. Intermediate for drug synthesis
TBAB plays a crucial role in drug synthesis, particularly in the fields of biopharmaceuticals and chemical pharmaceuticals
Synthesis of daggligin: TBAB catalyzes the deoxynucleophilic and electrophilic conversion of amide to generate α - aminophosphonate intermediate, which provides a key step for the synthesis of daggligin, a new anti diabetes drug.
Development of anti diabetes drugs: TBAB is used to catalyze the formation of carbon nitrogen bonds and carbon oxygen bonds, and promote the development of new hypoglycemic drugs.
3. Agricultural fungicides and insecticides
TBAB is used in agriculture to prevent and control crop diseases and pests, and to protect the healthy growth of crops
Fungicide: TBAB solution sprayed on the surface of leaves can inhibit fungal spore germination, prevent and control rice blast disease, wheat powdery mildew, etc.
Insecticides: TBAB is compounded with pyrethroid compounds to enhance contact and stomach toxicity against pests and reduce pesticide usage.

Innovative applications of water treatment and pollution remediation in the field of environmental protection

1. Adsorption and precipitation of heavy metal ions
TBAB removes heavy metal ions (such as Pb ² ⁺, Cd ² ⁺, Hg ² ⁺) from wastewater through chelation, forming insoluble oxalate or hydroxide precipitates:
Laboratory research: TBAB has a treatment efficiency of over 95% for lead containing wastewater (initial concentration of 100mg/L), with effluent lead concentration below 0.1mg/L (in compliance with the Comprehensive Wastewater Discharge Standard).
Industrial application: TBAB is combined with polyacrylamide to increase the particle size of sediment particles, accelerate sedimentation rate, and reduce wastewater treatment costs.
2. Degradation of organic pollutants
TBAB, as an electrolyte additive, promotes the degradation of organic pollutants in electrochemical oxidation methods
Dye wastewater treatment: The TBAB Na ₂ SO ₄ electrolysis system can efficiently degrade azo dyes (such as methyl orange), with a COD removal rate of over 90%.
Pesticide residue removal: TBAB catalyzes electrochemical reduction reactions to convert organic phosphorus pesticides (such as dichlorvos) into non-toxic products, reducing soil and water pollution.
3. Soil remediation and calcium ion regulation
In the improvement of saline alkali land, TBAB chelates Ca ² ⁺ in the soil, reduces exchangeable calcium content, and weakens the destructive effect of sodium ions on soil structure:
Field experiment: After applying TBAB, the soil pH value decreased by 0.5-1.0 unit, and the crop emergence rate increased by 15% -20%.
Mechanism study: TBAB forms soluble complexes with Ca ² ⁺, reducing the adsorption of calcium ions on soil particle surfaces and promoting sodium ion leaching.

Electrochemical and Materials Science: Preparation and Performance Optimization of Functional Materials

1. Electrolyte additives
TBAB, as an electrolyte additive, can improve the electrochemical performance of batteries and capacitors
Lithium ion battery: Tetrabutylammonium bromide powder added to the electrolyte can inhibit lithium dendrite growth and extend the battery cycle life.
Supercapacitors: TBAB organic solvent electrolysis system improves the energy density of double-layer capacitors, with a power density of up to 10kW/kg.
2. Preparation of nanomaterials
TBAB is used in nanotechnology and surface science to prepare highly conductive and stable nanocomposites:
Quantum dot synthesis: TBAB acts as a surface ligand to stabilize CdSe quantum dots, prevent aggregation, and improve fluorescence quantum yield.
Graphene modification: TBAB modifies the surface of graphene through π - π interactions, enhancing its interfacial bonding with polymers and improving the mechanical properties of composite materials.
3. Surfactants and emulsifiers
The quaternary ammonium salt structure of TBAB makes it an excellent surfactant for use in detergents, anti-static agents, and other fields
Detergent formula: TBAB reduces the surface tension of water, enhances stain removal ability, and has bactericidal effect.
Anti static coating: TBAB is compounded with polyvinyl alcohol to prepare a transparent anti-static film for electronic component packaging.

Other innovative application areas

1. Dye and pigment industry
TBAB is used to manufacture azo dyes, xanthene dyes, anthraquinone dyes, etc., and serves as a developer, wool mordant, and leather coloring agent:
Dye synthesis: TBAB catalyzes diazotization reaction to generate high-purity azo dyes with a color fastness of 4-5 levels.
Leather dyeing: TBAB forms complexes with metal ions to increase the adsorption capacity of dyes on leather fibers, resulting in uniform and full color.
2. Analytical chemical reagents
TBAB, as an ion pair reagent and polarographic analysis reagent, is used for chromatographic separation and electrochemical detection:
Ion pair chromatography: TBAB forms neutral ion pairs with acidic or alkaline analytes, improving chromatographic retention time and separation efficiency.
Polarographic analysis: TBAB serves as a supporting electrolyte to eliminate the influence of interfering ions and achieve high sensitivity detection of metal ions.
3. Paper waterproofing agent
TBAB is used in the paper manufacturing process to improve the water resistance and mechanical strength of paper
Coating process: TBAB is compounded with rosin adhesive to form a waterproof coating, reducing the water absorption of paper to below 5%.
Glue application effect: TBAB promotes the uniform distribution of rosin glue on the fiber surface, enhancing the tensile strength and flexural strength of the paper.


Preparation method of tetrabutylammonium bromide powder:
N (tri-n-butylamine): n (1-bromobutane)=1.0:1.1, reaction solvent ethyl acetate 20 mL, added to the autoclave, replaced the air in the autoclave with nitrogen three times, and filled with nitrogen until the pressure in the autoclave is 4 MPa, heated to 100 ℃ in the oil bath, and the reaction time is 14 h.
The product is subject to vacuum distillation to recover ethyl acetate and 1-bromobutane, and the mixture of petroleum ether and 1-bromobutane is added to disperse the product after distillation. The mixture of petroleum ether and 1-bromobutane with a mass ratio of 1.0:1.3 is added three times, and then washed and filtered. Put the filtered solid material into the vacuum drying oven to dry to constant weight, and then the product can be obtained, with the yield of about 83.93% and purity of about 98.96%.
Reaction mechanism:
The TBAB synthesis reaction appears to be a secondary reaction, and the reaction may be a SN2 reaction. Tributylamine has a lone pair of electrons, which acts as a nucleophilic reagent to attack 1-bromobutane. When the nucleophilic reagent gradually forms a new bond with the saturated carbon atom of bromobutane, the leaving group Br is gradually pushed out of the saturated carbon atom to form an activated substance and then converted into a product. Because of the production of the activated substance, the order of the process increases, thus △ S0<0. The activation energy of the whole reaction is large. Increasing the reaction temperature can help the reaction to proceed faster.
![]()
Tetrabutylammonium bromide Chemical properties:
In 1951, Buckles and others prepared TBABr3 by dissolving tetrabutylammonium bromide in brominated carbon tetrachloride solution. The reaction can be completed in a short time with a yield of 91%. TBABr3 can also be obtained by passing the bromine vapor through the tetrabutylammonium bromide solid without solvent and contacting the vapor with the solid for several hours, with a yield of 84% .
![]()
In 1987, Kajigaeshi et al. dissolved tetrabutylammonium bromide in the aqueous solution of sodium bromate and added hydrobromic acid to the solution to prepare TBABr3 with a yield of 95%, which is more convenient to operate and avoids the use of simple bromine.
![]()
Weigh a certain amount of quaternary ammonium salt and add it into the reaction vessel, add a certain amount of water and silver oxide, and then stir for 1h in the next instant. Take samples for centrifugation and test whether the supernatant still contains halogen ions. If it still contains halogen ions, add silver oxide until no halogen ions are detected. Filter the reaction solution, and the reverse phase is the aqueous solution of quaternary ammonium base. In order to prevent the quaternary ammonium base in the solution from being oxidized by carbon dioxide in the air, filtration needs to be carried out in nitrogen.
![]()
Mix the methanol solution of tetrabutylammonium bromide with the methanol solution of potassium hydroxide, and react for 0.5h. The mixture obtained after reaction is filtered, and part of anhydrous methanol as solvent is removed by atmospheric distillation. Then, add pure water and continue atmospheric distillation to remove methanol and trin-butylamine produced by micro decomposition. Finally, the obtained concentrate is transferred to the crystallizer, cooled, crystallized and filtered under the protection of quicklime absorption device. The filtered product is dried in a vacuum drying oven at 80 ℃ to obtain white crystal product.
![]()
Gel type strong basic anion exchange resin 201 × 4. After pretreatment, it is loaded into the column, and a certain concentration of tetrabutylammonium bromide solution is prepared for ion exchange to obtain tetrabutylammonium hydroxide aqueous solution. The ion exchange resin is regenerated with NaOH solution and recycled. The exchangeable ion OH - carried by the resin functional group is used for ion exchange with tetrabutylammonium bromide solution, Br - is adsorbed on the resin, and OH - enters the solution.
![]()
Tetrabutylammonium bromide is decomposed into 52% and 92% tributylamine in concentrated NaOH aqueous solution at 60 or 100 ℃ after 7 hours.
![]()
In gas chromatography, the minimum temperature for the complete decomposition of tetrabutylammonium bromide into salt in aqueous solution is 300 ℃.
![]()
The combination of 2-iodoylbenzoic acid (IBX) and tetrabutylammonium bromide can be used for the decarboxylation and bromination of cinnamic acid. Some metal halides and other phase transfer catalysts can be used as halogenation reagents. Acetonitrile - water (2:1, v/v) is the best solvent system. Under the condition of strong electron absorption, better yields can be obtained.

Under heating conditions, N-alkylation of indole can take place between tetrabutylammonium bromide and 3-indole.

Tetrabutylammonium bromide can react with enolate to form alkylation product.

Test method:
1. Titration:
Accurately weigh about 1.0 g of sample, dissolve it in a 250 mL conical flask, and add 50 mL of water. Add 0.05 g of sodium bicarbonate and 0.2 mL of eosin reagent solution. Titrate with 0.1 g/L silver nitrate solution until the first discoloration. 1 mL 0.1 g/L silver nitrate solution is equivalent to 0.03224 g tetrabutylammonium bromide.
TBAB content (%)=volume of consumed standard solution (mL) × 0.03224 ÷ 1 g (sample weight) × 100%
2. Polarography:
Cathode: drop mercury electrode
Anode: Pt wire
Prepare 1.0 M solution (32.24 g tetrabutylammonium bromide in 100 mL of water), and add 0.2 mL of 1.0 M tetrabutylammonium bromide powder. Deoxidize with high-purity nitrogen until the minimum background current is reached (about 5 minutes). By using the condition that the modulation amplitude is 50 mV with a decrease of 1 s, the scanning range is from - 1.2 to - 2.6 V with a sensitivity of 0.5 µ A at 0.01 V/s. If the current is observed to be greater than 0.05 µ A, it means that the material meets the requirements without impurities.
Frequently Asked Questions
What is tetrabutylammonium acetate used for?
+
-
Tetrabutylammonium acetate is an organic compound commonly used as a catalyst and solvent. It can be used to catalyze or promote reactions in certain organic reactions, and is widely used in batteries, solar cells and pigments.
Is tetrabutylammonium bromide toxic?
+
-
CONSIDERED A HAZARDOUS SUBSTANCE ACCORDING TO OSHA 29 CFR 1910.1200. Harmful if swallowed. Irritating to skin. Risk of serious damage to eyes.
What does tetrabutylammonium bromide do?
+
-
Tetrabutylammonium Bromide used in many
Phase-Transfer Catalyst: TBAB is widely used as a phase-transfer catalyst in organic synthesis reactions. It facilitates the transfer of reactants between immiscible phases, such as aqueous and organic phases, thereby accelerating reaction rates and improving yields.
What is Tetramethylammonium bromide used for?
+
-
Tetramethylammonium bromide (TMABr) is a quaternary ammonium salt that is widely used as a phase transfer catalyst. Absolute viscosities of its aqueous solutions have been measured at various temperatures (20,25 and 30°C).
Hot Tags: tetrabutylammonium bromide powder cas 1643-19-2, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, N N N Trimethylethylenediamine, 2 5 Dihydroxybenzaldehyde, 2 6 Pyridinedicarboxylic acid, Additive, gs 441524 remdesivir, BENZENE D6









