Seraspenide, also known as goralatide, is an endogenous tetrapeptide with significant biological activity, widely distributed in various tissues and body fluids. It is hydrolyzed by its precursor thymosin β 4 by prolyl oligopeptidase (POP). The concentration of goratide in the blood is usually within the nanomolar range.In terms of pharmacokinetics, goralatide is quickly degraded after intravenous administration, with a half-life of only 4 to 5 minutes. It is eliminated from the plasma through two main mechanisms: angiotensin-converting enzyme (ACE)-mediated hydrolysis and glomerular filtration.
AC-SER-ASP-LYS-PRO-OH is a multifunctional physiological regulator that possesses various biological activities. Early studies showed that goralatide could inhibit the activity of hematopoietic stem cells by preventing them from entering the S phase and keeping them in the G0 phase. More recently, it has been discovered that goralatide can enhance epidermal re-implantation ability and accelerate wound healing in damaged avascular epidermal grafts by promoting angiogenesis. Additionally, goralatide can inhibit the differentiation of bone marrow stem cells into macrophages stimulated by macrophage growth medium (MGM), thus exhibiting anti-inflammatory effects.
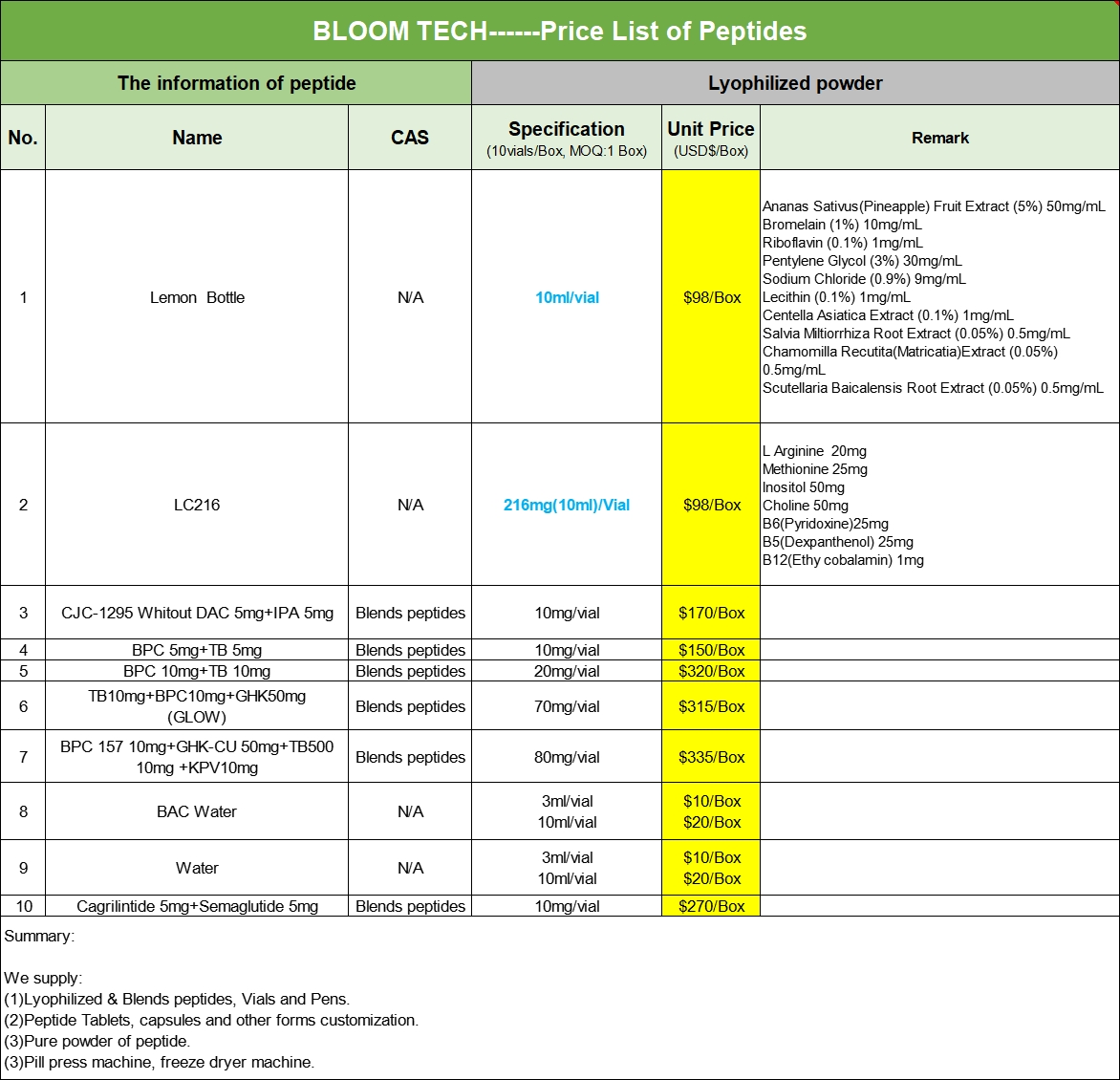
Our Products Form





Seraspenide\AC-SER-ASP-LYS-PRO-OH COA
 |
||
| Certificate of Analysis | ||
| Compound name | Seraspenide\AC-SER-ASP-LYS-PRO-OH | |
| Grade | Pharmaceutical grade | |
| CAS No. | 127103-11-1 | |
| Quantity | 62g | |
| Packaging standard | PE bag+Al foil bag | |
| Manufacturer | Shaanxi BLOOM TECH Co., Ltd | |
| Lot No. | 202601090088 | |
| MFG | Jan 9th 2026 | |
| EXP | Jan 8th 2029 | |
| Structure |
|
|
| Item | Enterprise standard | Analysis result |
| Appearance | White or almost white powder | Conformed |
| Water content | ≤5.0% | 0.44% |
| Loss on drying | ≤1.0% | 0.58% |
| Heavy Metals | Pb≤0.5ppm | N.D. |
| As≤0.5ppm | N.D. | |
| Hg≤0.5ppm | N.D. | |
| Cd≤0.5ppm | N.D. | |
| Purity (HPLC) | ≥99.0% | 99.90% |
| Single impurity | <0.8% | 0.12% |
| Total microbial count | ≤750cfu/g | 500 |
| E. Coli | ≤2MPN/g | N.D. |
| Salmonella | N.D. | N.D. |
| Ethanol (by GC) | ≤5000ppm | 600ppm |
| Storage | Store in a sealed, dark, and dry place below -20°C | |
|
|
||
|
|
||
| Chemical Formula | C20H33N5O9 |
| Exact Mass | 487.23 |
| Molecular Weight | 487.51 |
| m/z | 487.23 (100.0%), 488.23 (21.6%), 489.23 (2.2%), 489.23 (1.8%), 488.22 (1.8%) |
| Elemental Analysis | C, 49.27; H, 6.82; N, 14.37; O, 29.54 |

Hematoprotective Agent in Tumor Chemotherapy and Radiotherapy
Protection against Chemotherapy-Induced Myelosuppression
Myelosuppression is the most common dose-limiting toxicity of tumor chemotherapy. Chemotherapeutic agents such as cytarabine, ifosfamide, cyclophosphamide and carboplatin indiscriminately damage highly proliferative hematopoietic stem cells, leading to neutropenia, thrombocytopenia and anemia. This elevates the risk of infection and bleeding, forces chemotherapy dose reduction or treatment interruption, and seriously compromises therapeutic efficacy.
As a specific cell cycle inhibitor of normal hematopoietic stem cells, seraspenide precisely blocks the transition of hematopoietic stem cells from the G0/G1 phase to the S phase, maintaining them in a quiescent state. It thereby protects these cells from being killed by chemotherapeutic agents targeting proliferating cells, without interfering with the cell cycle of tumor cells which lack the corresponding target. It achieves the dual goal of protecting normal cells without weakening chemotherapy efficacy.Clinical studies have shown that among 53 cancer patients receiving single-agent chemotherapy with cytarabine or ifosfamide, it administered at a dose of 200 μg/m²/d starting 24 hours before chemotherapy for 5 consecutive days significantly elevated the nadir values of peripheral blood leukocytes, neutrophils and platelets.
It shortened the duration of myelosuppression and reduced the incidence of severe agranulocytosis (neutrophils < 0.5×10⁹/L), with no obvious toxic side effects and no increased risk of tumor recurrence. Animal experiments further confirmed that it raised the survival rate of mice receiving lethal-dose chemotherapy by more than 40% and accelerated the recovery rate of bone marrow hematopoietic stem cell count by twofold, providing a reliable safety guarantee for the implementation of high-dose and dose-dense chemotherapy regimens.
Protection against Radiotherapy-Induced Hematological and Mucosal Injury
Radiotherapy, especially total body irradiation, pelvic irradiation and bone marrow irradiation, directly damages the bone marrow microenvironment and the DNA of hematopoietic stem cells, resulting in persistent bone marrow failure. Meanwhile, it injures mucosal epithelial cells of the digestive and respiratory tracts, triggering complications such as mucositis, diarrhea and secondary infection.It exerts protective effects through dual mechanisms: firstly, it keeps hematopoietic stem cells quiescent to reduce DNA damage induced by radiotherapy.
Secondly, it suppresses radiotherapy-triggered oxidative stress and inflammatory responses, lowers reactive oxygen species (ROS) levels, and decreases DNA fragmentation and cell apoptosis.
In total body irradiation (TBI) conditioning prior to hematopoietic stem cell transplantation (HSCT), it markedly reduced the incidence of post-transplant bone marrow failure, accelerated hematopoietic reconstitution, and shortened the engraftment time of neutrophils and platelets.
For oral mucositis caused by radiotherapy for head and neck tumors, local administration of it promotes the repair of mucosal epithelial cells, alleviates pain and ulcer severity, and reduces the risk of eating difficulty and secondary infection. Its mechanism is related to inhibiting the release of inflammatory factors (TNF-α, IL-6) and facilitating the proliferation and differentiation of mucosal stem cells.

Adjuvant Therapy for High-Dose Chemotherapy and Hematopoietic Stem Cell Transplantation
High-dose chemotherapy combined with autologous hematopoietic stem cell transplantation (ASCT) is a curative treatment for hematological malignancies such as lymphoma and multiple myeloma. Nevertheless, high-dose chemotherapy often causes severe myelosuppression, accompanied by delayed hematopoietic reconstitution and a high risk of graft-versus-host disease (GVHD) after transplantation.
The application of it in ASCT is divided into two stages: administration before high-dose chemotherapy protects endogenous hematopoietic stem cells and alleviates chemotherapy-induced damage; administration after transplantation promotes the homing, proliferation and differentiation of infused hematopoietic stem cells to accelerate hematopoietic reconstitution. Meanwhile, it inhibits the activation of allogeneic immune cells and reduces the incidence of acute GVHD.

Cardiovascular and Renal Diseases: Anti-Fibrotic and Organ-Protective Agent
Intervention on Myocardial Fibrosis in Hypertension and Heart Failure
Myocardial fibrosis is a core pathological alteration in hypertension and heart failure, characterized by excessive proliferation of myocardial fibroblasts and massive deposition of type Ⅰ and type Ⅲ collagen. This leads to myocardial stiffness, diastolic dysfunction and arrhythmia, eventually progressing to heart failure.
Seraspenide exerts myocardial protection via multi-target anti-fibrotic mechanisms. First, it inhibits the proliferation and activation of myocardial fibroblasts, blocks the Smad2/3 signaling pathway-the core pathway of fibrosis-and reduces collagen synthesis and deposition.
Second, it suppresses angiotensin Ⅱ (Ang Ⅱ)-induced inflammatory responses, decreases levels of inflammatory factors such as TNF-α and IL-1β, and mitigates myocardial inflammatory injury. Third, it promotes myocardial microangiogenesis, improves myocardial blood and oxygen supply, and reverses myocardial remodeling.
In hypertensive rat models, it at 1 mg/kg/d administered continuously for 8 weeks significantly reduced myocardial collagen content by over 40%, improved myocardial diastolic function, and alleviated left ventricular hypertrophy without affecting blood pressure, indicating its anti-fibrotic effect is independent of hypotensive action.


In patients with heart failure, combined treatment of it and ACE inhibitors (ACEI) further downregulated the levels of myocardial fibrosis biomarkers (N-terminal propeptide of type Ⅰ procollagen, N-terminal propeptide of type Ⅲ procollagen), improved left ventricular ejection fraction (LVEF), and reduced hospital readmission and mortality rates.
Prevention and Treatment of Renal Fibrosis in Diabetic Nephropathy and Chronic Kidney Disease
Renal fibrosis is the common terminal pathological feature of chronic kidney diseases including diabetic nephropathy, hypertensive renal damage and chronic glomerulonephritis, manifesting as glomerulosclerosis and tubulointerstitial fibrosis, which ultimately develop into end-stage renal disease (ESRD).
As an endogenous anti-fibrotic peptide, it inhibits the epithelial-mesenchymal transition (EMT) of renal mesangial cells and renal tubular epithelial cells into myofibroblasts, reduces extracellular matrix (ECM) deposition, and blocks the progression of renal fibrosis.In diabetic nephropathy rat models, it markedly decreased urinary albumin excretion rate by more than 50%, alleviated glomerular mesangial proliferation and tubulointerstitial fibrosis, preserved podocyte integrity, reduced the loss of nephrin protein, and delayed the decline of renal function.
Clinical studies have shown that it combined with ACEI/ARB drugs significantly lowered serum creatinine and blood urea nitrogen levels, reduced proteinuria, and slowed the decline of estimated glomerular filtration rate (eGFR) in chronic kidney disease patients, with good safety and no obvious adverse reactions. Moreover, it can be applied to the prevention and treatment of chronic allograft nephropathy after renal transplantation, inhibiting interstitial fibrosis of the grafted kidney and prolonging graft survival time.


I. Solid-Phase Synthesis (Mainstream Industrial Process)
Seraspenide is an N-acetylated tetrapeptide (Ac-Ser-Asp-Lys-Pro). Its industrial production mainly adopts Fmoc solid-phase peptide synthesis (SPPS) with Rink Amide resin as the carrier. The synthesis proceeds by stepwise coupling from the C-terminus to the N-terminus, featuring controllable whole-process reaction and high product purity.
Resin activation and amino acid coupling: The resin is first swollen with dimethylformamide (DMF), and the Fmoc protecting group is removed using 20% piperidine/DMF solution. Amino acids including Fmoc-Pro-OH, Fmoc-Lys(Boc)-OH, Fmoc-Asp(OtBu)-OH and Fmoc-Ser(tBu)-OH are sequentially coupled in the order of Pro→Lys→Asp→Ser. HBTU/HOBt/DIPEA is applied as the condensation agent in each step, with the reaction carried out at room temperature for 30–60 minutes to ensure complete coupling.
N-terminal acetylation and side-chain deprotection: After the assembly of the tetrapeptide skeleton, the N-terminal amino group is acetylated with acetic anhydride/DMF to obtain Ac-Ser(tBu)-Asp(OtBu)-Lys(Boc)-Pro-resin. Trifluoroacetic acid (TFA) is then used to cleave the resin and remove side-chain protecting groups including tBu and Boc. The mixed system of TFA/water/triisopropylsilane (95:3:2) reacts at room temperature for 2 hours, and the crude peptide is collected by diethyl ether precipitation.
Purification and lyophilization: The crude product is purified by reversed-phase high-performance liquid chromatography (RP-HPLC) on a C18 column with acetonitrile-water containing 0.1% TFA as the mobile phase for gradient elution. The main peak fraction is collected and lyophilized to obtain a white powdered pure product, with an overall yield of approximately 50%–60% and purity no less than 98%.
II. Liquid-Phase Synthesis (Laboratory Preparation)
Liquid-phase synthesis is suitable for small-batch preparation. It adopts stepwise condensation via the mixed anhydride method, offering flexible synthetic routes but requiring multiple purification procedures.
Fragment construction: The C-terminal dipeptide Boc-Lys(Boc)-Pro-OH and the N-terminal dipeptide Ac-Ser-Asp(OBzl)-OH are synthesized first. The side chains are protected with Boc and benzyl (Bzl) groups respectively to avoid side reactions.
Fragment condensation: The two dipeptide fragments react under DCC/HOBt condensation conditions to generate the fully protected tetrapeptide Ac-Ser-Asp(OBzl)-Lys(Boc)-Pro-OH.
Deprotection and purification: Catalytic hydrogenation with palladium on carbon (Pd/C) is used to remove the Bzl group, and TFA is applied to eliminate the Boc group. The product is dialyzed, purified by RP-HPLC and lyophilized, achieving a yield of about 35%–45% and purity no less than 95%.
III. Enzymatic Synthesis (Green Process)
Enzymatic synthesis prepares it by specific cleavage of thymosin β4 (Tβ4) with prolyl oligopeptidase (POP), characterized by mild reaction conditions and high stereoselectivity.
Tβ4 is dissolved in phosphate buffer solution at pH 7.4, followed by the addition of POP enzyme and incubation at 37 ℃ for 12–24 hours to produce AcSDKP via enzymatic hydrolysis.
The reaction solution is treated by ultrafiltration to remove enzymes and macromolecular impurities, then purified by RP-HPLC and lyophilized. The yield reaches approximately 40%–50%. The product retains high natural biological activity and is suitable for the research and development of high-end pharmaceutical formulations.


AC-SER-ASP-LYS-PRO-OH is a bioactive peptide with potential medicinal value, so it is crucial to study its pharmacokinetics. Pharmacokinetic studies typically include processes such as drug absorption, distribution, metabolism, and excretion in the body, as well as interactions between drugs and related molecules in the organism. However, there may still be relatively few studies on its pharmacokinetics, as the study of Gorelenide, as a novel bioactive peptide, is still in its early stages.
1. Absorption: Absorption usually occurs in the digestive tract and can be administered orally or by injection. The speed and degree of absorption may be influenced by various factors, such as drug form, route of administration, and intestinal absorption conditions.
2. Distribution: The distribution within the body is influenced by biological barriers and tissue specificity. It may be distributed to various tissues and organs in the bloodstream, and may have specific affinity in some tissues.
3. Metabolism: It may undergo metabolic processes in the body, including enzyme mediated degradation and modification. The properties and metabolic pathways of metabolites may affect the pharmacological and pharmacokinetic properties of Gorelelutide.
4. Excretion: Excretion is usually carried out through the kidneys and/or liver, and may be excreted from the body in the form of urine and/or feces. The excretion rate and pathway may be influenced by various factors, including kidney function, liver function, etc.
5. Drug interactions: May interact with other molecules in the body (such as receptors, proteins, etc.), affecting their pharmacological and pharmacokinetic properties. These interactions may affect the absorption, distribution, metabolism, and excretion processes of Goreletin.
Hot Tags: SERASPENIDE / AC-SER-ASP-LYS-PRO-OH CAS 127103-11-1, suppliers, manufacturers, factory, wholesale, buy, price, bulk, for sale, N2 N4 di 1 1 biphenyl 4 yl 9 9 dimethyl N2 N4 diphenyl 9H fluorene 2 4 diamine, 7 3 pyren 1 yl phenyl dibenzo c h acridine, 2 3 diphenyl 6 1 phenyl 1H benzo d imidazol 2 yl quinoxaline, 2 4 Quinolinediol, tris 4 benzo d oxazol 2 yl phenyl amine, Cosmetic






















